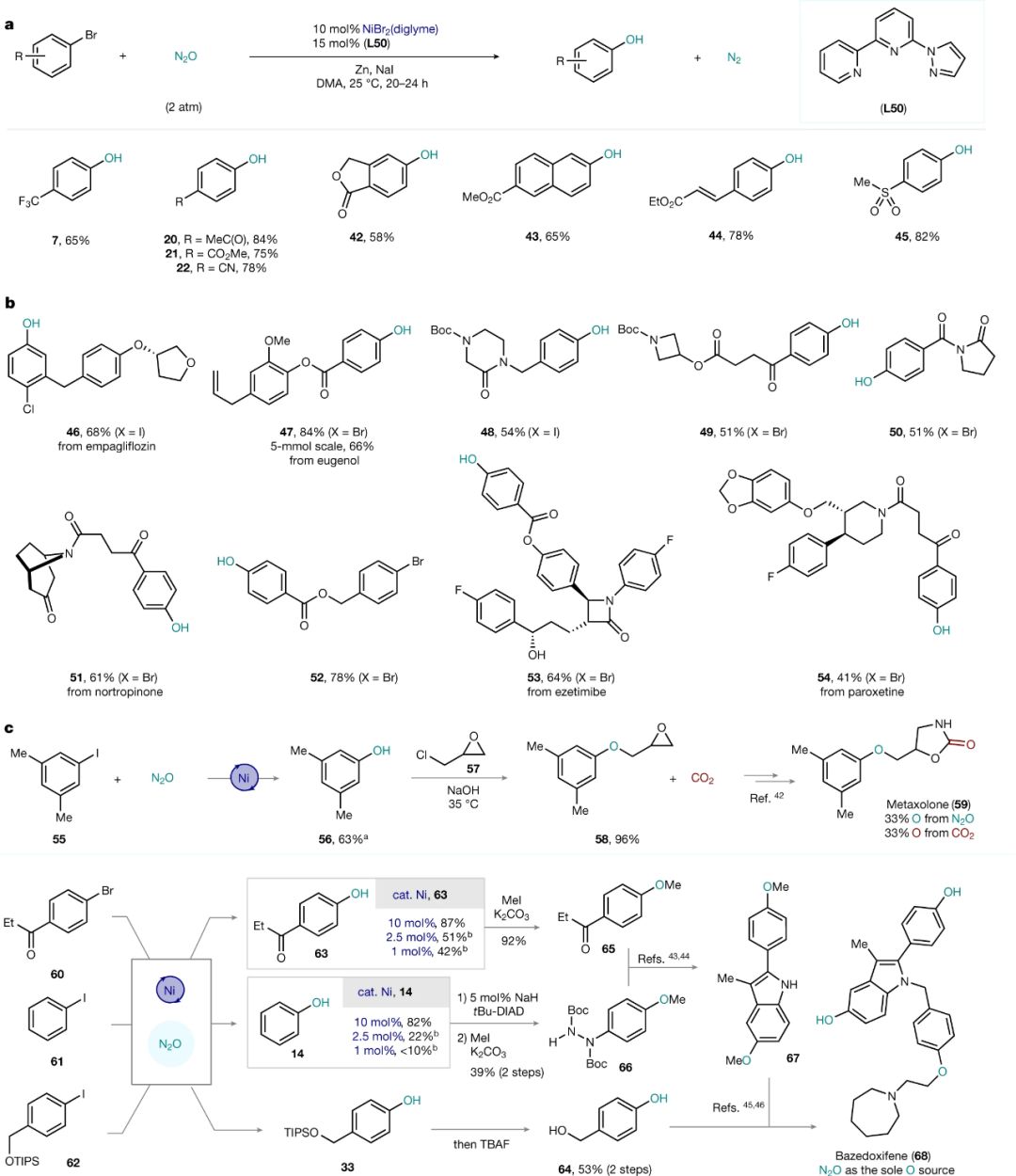
利用卤代芳烃制备相应的酚,传统的方法是利用Ullmann偶联反应和Buchwald型钯催化的C-O键形成反应。但这两种方法通常重复性较差,另外都要在碱性条件下进行,通常需要加热到较高温度。对于一些对碱敏感的底物,这一转化会受到限制。
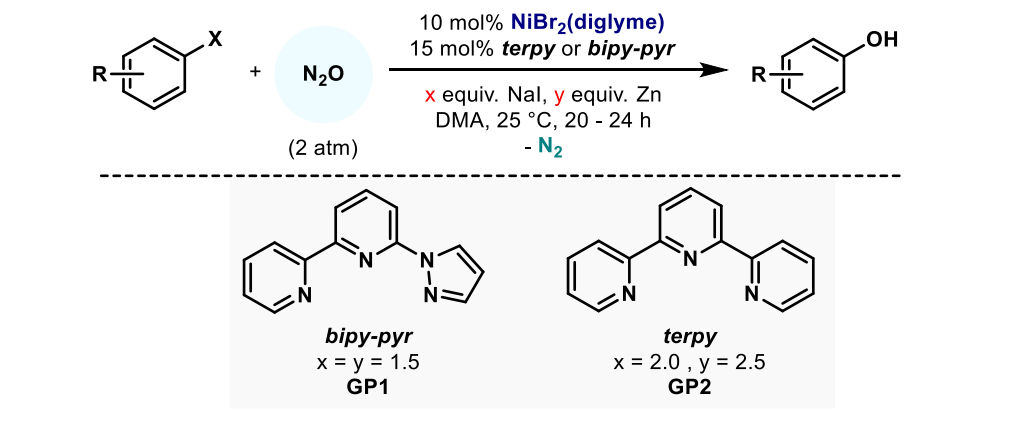
近期,德国马克思·普朗克煤炭研究所(Max-Planck-Institut fu r Kohlenforschung)的Josep Cornella教授团队报道一种利用亲电型N2O作为氧源,Ni-联吡啶三齿配体催化下,将卤代芳烃氧化为相应酚的方法。相关成果发表在顶级期刊Nature上【Nature 604, 677–683 (2022)】。此方法操作简便,条件温和,选择性高,能够适用于对碱敏感的底物,官能团耐受度高。
反应中N2O在温和条件下(室温下, 1.5–2 bar N2O)插入Ni-C键,得到酚并放出氮气。此方法和现存的通过还原消除形成C-O键路径不同, C(sp2)‒O 形成过程是通过有机金属Baeyer–Villiger型机理进行的,N2O先和金属中心络合,亲电的氧最终插入到金属M和sp2-C中间,同时放出氮气,整个过程中金属中心的价态不变,因此还要加入其他的还原剂实现催化循环,Ni由于有多个价态,可以通过单电子转移实现价态转换,这正是选用Ni用于此反应的原因。
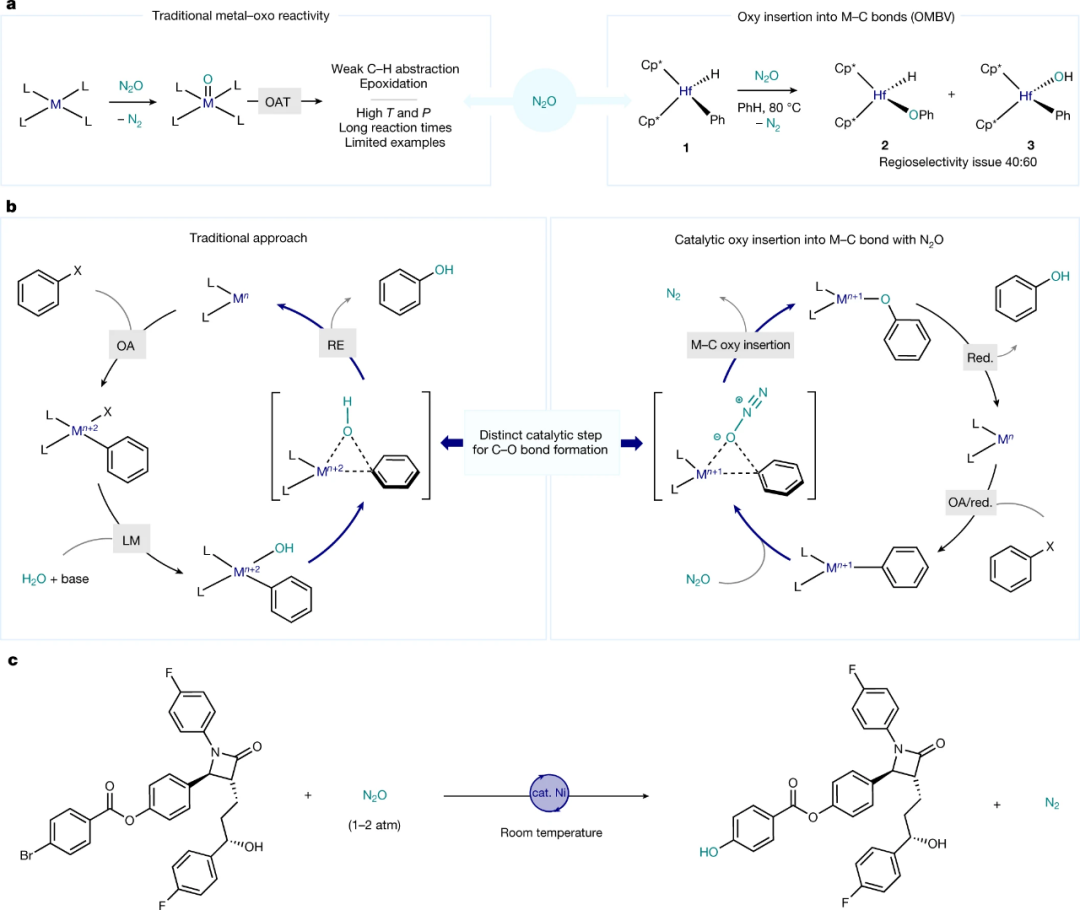
作者发现利用Zn/NaI作为还原剂效果最佳,在此基础上筛选了70多个联吡啶类配体发现三联吡啶(L18)和6-吡唑基-2,2'-联吡啶 (L50) 产率最高。
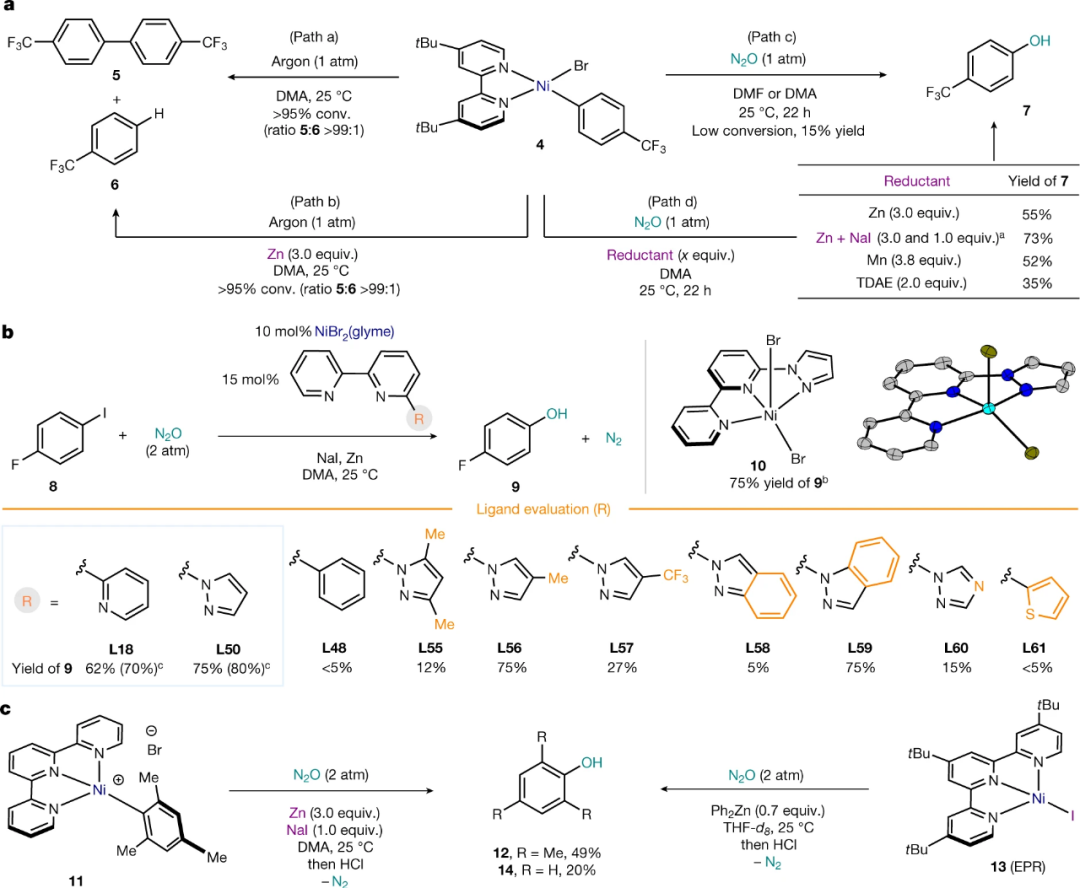
利用筛选出的反应条件,进行底物适用范围验证,发现含有其他卤代的芳基碘代物也可以顺利生成相应的酚,吸电子取代基和给电子取代的芳基碘代物都可以高产率得到相应的酚。邻位具体较大位阻的取代基会明显影响产率。
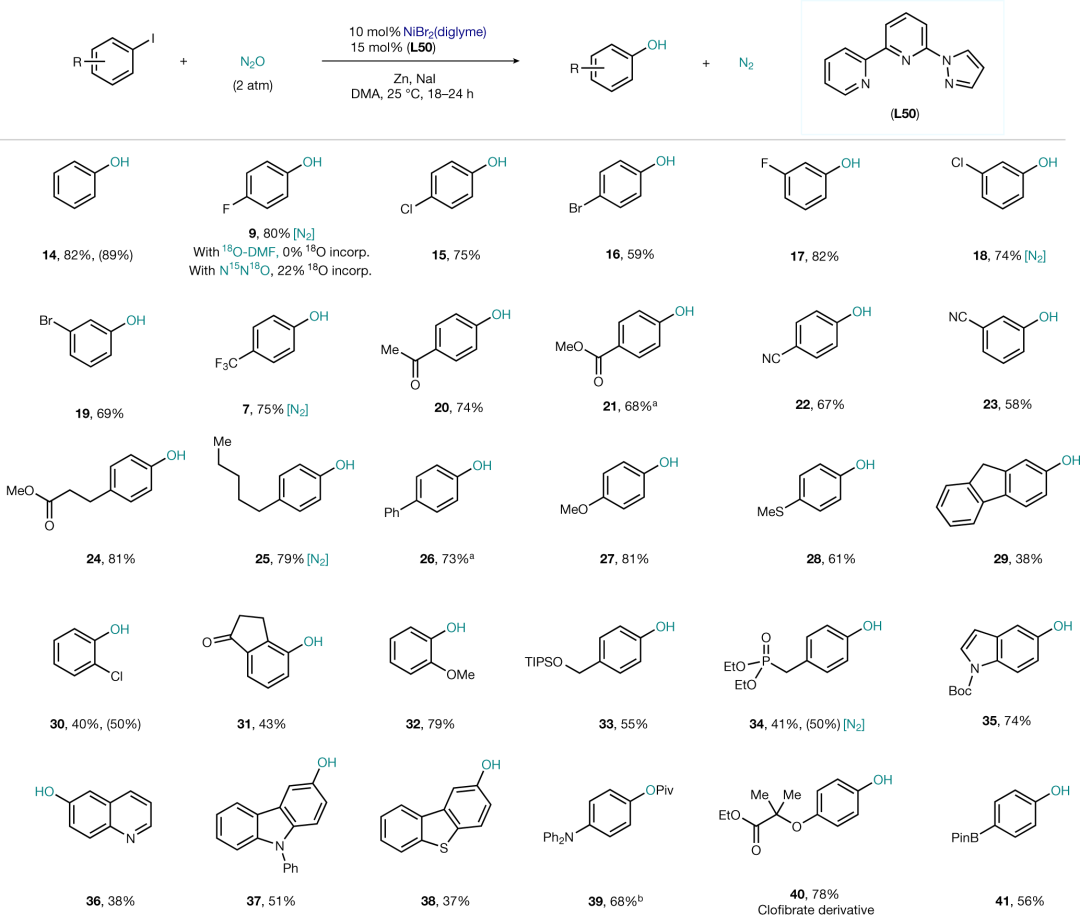
含有吸电子取代基的溴代芳烃可以高产率得到相应的酚,一些结构相对复杂的医药中间体也能顺利反应。
反应操作
General procedure GP1: (using bipy-pyr-L50)
For the scope, oven- and heatgun-dried pressure Schlenks with Teflon screw-cap and equipped with a Teflon-coated stir bar were used. These Schlenks were brought into an argon filled glovebox, along with a heatgun-dried vial. Dried DMA was introduced into the vial. NiBr2·diglyme (7.1 mg, 0.020 mmol, 10 mol%), bipy-pyr L50 (6.7 mg, 0.030 mmol, 15 mol%), NaI (45 mg, 0.30 mmol, 1.5 equiv), aryl halide (if solid, 0.20 mmol, 1.0 equiv), and zinc (20 mg, 0.30 mmol, 1.5 equiv) were introduced in the pressure Schlenk. Then, outside the glovebox, using a T-connection, the Schlenk was evacuated and refilled with N2O. This procedure was repeated three times. Then, under 1.5 atm of N2O, the Schlenk was opened and DMA (0.5 mL) was added using a syringe while stirring. (Stirring is important to avoid formation of agglomerates.) If the aryl halide was liquid, it was added at that time using the adapted size of Hamilton syringe. The Schlenk was then closed, and the pressure of N2O was increased to 1.9 – 2.0 atm. The reaction mixture was kept stirring at 1000 rpm, for 20 to 24 h, at RT. After the reaction, the Schlenk was carefully opened, and the reaction was first diluted using 2 mL of Et2O or EtOAc depending on the solubility of the expected product. The mixture was quenched first with water (the quenching can be exothermic) followed by HCl (1 M), and extracted 3 times with Et2O (12 to 15 mL). The combined organic layers were washed with brine, dried over MgSO4, filtered and concentrated under reduced pressure. The crude was purified by column chromatography over silica gel using gradient of solvents.
General procedure 2: (terpy conditions)
The sole modifications for these conditions are highlighted here: NiBr2·diglyme (7.1 mg, 0.020 mmol, 10 mol%), terpy L18 (7.0 mg, 0.030 mmol, 15 mol%), NaI (60 mg, 0.40 mmol, 2.0 equiv), aryl halide (if solid, 0.20 mmol, 1.0 equiv), and zinc (32.7 mg, 0.500 mmol, 2.5 equiv) were introduced in the pressure Schlenk.
参考资料
Le Vaillant, F., Mateos Calbet, A., González-Pelayo, S.et al.Catalytic synthesis of phenols with nitrous oxide. Nature 604, 677–683 (2022). https://doi.org/10.1038/s41586-022-04516-4
